For many years, longevity science has relied heavily on molecular proxies: NAD⁺ levels, inflammatory markers, signaling pathways, or mechanistic hypotheses derived from cellular or animal models. Those tools are valuable; we use them ourselves.
But at some point, a longevity intervention must answer a harder question: Does it measurably influence how a human organ system functions?
In our recently completed university-run randomized, double-blind, placebo-controlled clinical trial in 61 healthy adults aged 40+, we chose to measure endpoints that reflect real-world vascular physiology. Not molecules in isolation or short-term signaling shifts, but rather validated measures of endothelial function, arterial flexibility, and hemodynamics.
The full manuscript, which has now been submitted for peer review, details the methodology and statistical approach. Here, I’d like to step back and examine what the results mean in the context of human longevity research, especially in relation to other nutritional and lifestyle interventions.
What We Measured And Why It Matters
The primary endpoint was flow-mediated dilation (FMD), a widely used non-invasive measure of endothelial function. Secondary endpoints included carotid-femoral pulse wave velocity (PWV) and systolic blood pressure (SBP), along with SCORE2/SCORE2-OP estimated 10-year cardiovascular risk. We also measured metabolic markers.
We selected cardiovascular physiology as the first system to evaluate deliberately. Cardiovascular disease remains the #1 age-related cause of death globally and is responsible for roughly 1.5–2x more deaths than cancer each year. Vascular dysfunction emerges early, influences organ perfusion throughout the body, and is tightly linked in epidemiological research to both healthspan and lifespan. If a longevity intervention meaningfully affects aging biology, cardiovascular function is one of the most relevant and measurable places to begin.
These metrics are routinely used in vascular and aging research and have been associated in epidemiological studies with long-term cardiovascular outcomes, and more broadly, healthspan and lifespan.
Importantly, these measures are difficult to move in generally healthy, normotensive adults, and even more difficult to move simultaneously.
The Observed Effects in Context
1. Endothelial Function (FMD)
At 6 months:
- FMD increased by +2.6% (±2.0%) in the NOVOS Core group
- Placebo changed by −0.1%
- Baseline-adjusted between-group difference: +2.9% (95% CI 2.1–3.8)
- The results exhibited a strong statistical significance (p = 0.0001)
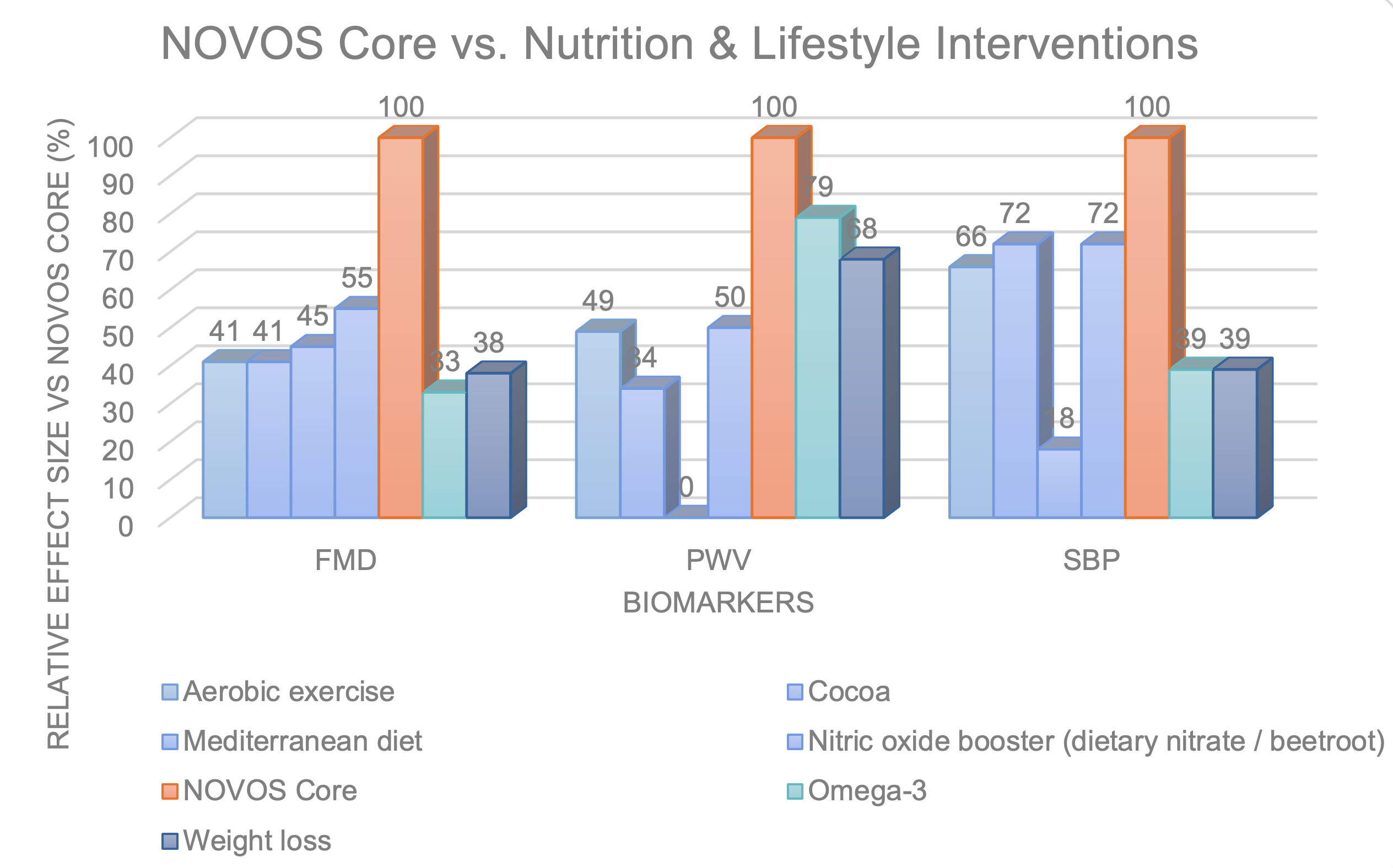
How does NOVOS Core compare with other FMD interventions?
Meta-analyses of cocoa flavanols, nitric oxide boosters like beetroot nitrate and l-citrulline / l-arginine, and tea polyphenols typically report sustained improvements in the range of ~0.8–1.5 percentage points in healthy or mildly at-risk adults.

Structured exercise interventions often fall in the ~1–2% range depending on population and duration.
A sustained ~2.9% between-group difference over six months, measured ~24 hours after the last dose (participants were fasted in the NOVOS Core study), is therefore on the higher end of what is typically reported for nutritional strategies in similar populations.
Equally notable is that FMD improved acutely at 1 -2 hours and remained improved after 6 months of daily use. This suggests both short-term endothelial responsiveness and sustained vascular adaptation.

Individual flow-mediated dilation (FMD) data in NOVOS Core (blue) and Placebo (orange) groups at Visit 1 and after chronic daily ingestion of intervention (Visit 2). (B) Changes of FMD at Visit 2 relative to Visit 1 with mean changes in groups. Error bars= SD, * p < 0.0001.
In observational meta-analyses, each 1% higher FMD has been associated with a lower rate of future cardiovascular events. That does not mean this product reduces events (this study was not designed or powered to measure clinical outcomes), but it reinforces that FMD is a clinically relevant functional biomarker.
(See exhibit for table of results and study references).
2. Arterial Flexibility (PWV)
At 6 months:
- PWV decreased by −0.78 m/s in the NOVOS Core group
- Increased by +0.56 m/s in placebo
- Between-group difference: −1.18 m/s (95% CI −2.0 to −0.36)
- The results exhibited a strong statistical significance (p = 0.006)
Carotid–femoral PWV is widely considered the gold-standard non-invasive marker of arterial flexibility and vascular aging.
In longitudinal population studies, PWV typically increases by roughly ~1 m/s per decade of aging, though exact rates vary by cohort. The magnitude observed here over six months is therefore comparable in scale to what epidemiologic data associate with multi-year age-related vascular change.
How does NOVOS Core compare with other PWV interventions?
Lifestyle strategies can improve arterial flexibility, but the magnitude and consistency vary:
- High-intensity interval training (HIIT) meta-analyses often report PWV reductions in the range of ~0.5–0.7 m/s in older adults.
- Aerobic exercise training programs show modest improvements, typically under ~0.6 m/s depending on baseline flexibility.
- The DASH heart-health diet, particularly when combined with weight management, can produce reductions in arterial flexibility in certain populations and comes the closest to this study’s results (1.07 m/s).
- Severe weight loss in overweight or metabolically unhealthy individuals can reduce PWV meaningfully (0.8 m/s), though the magnitude often depends on the degree of weight reduction and baseline risk.

Interestingly, the Mediterranean diet, often regarded as one of the most effective dietary patterns for longevity, did not significantly improve PWV in a randomized study of healthy adults aged 65–79 years (NU-AGE trial) over one year.
This does not diminish the value of the Mediterranean diet, which has strong associations with long-term cardiovascular outcomes. It simply highlights that improving arterial flexibility in already healthy older adults is not easy, even with high-quality dietary interventions.
In that context, NOVOS Core’s −1.18 m/s between-group difference over six months in generally healthy adults represents an effect size that is at least comparable to, and in many cases larger than, many lifestyle or nutritional interventions studied in similar populations.
Again, this does not imply reversal of aging or disease prevention; more research is necessary before arriving at that conclusion. It means that a validated physiological marker of vascular flexibility moved in a direction consistent with improved arterial flexibility, within a randomized controlled human trial.
(See exhibit for table of results and study references).
3. Systolic Blood Pressure (SBP)
At 6 months:
- Systolic Blood Pressure changed by −8.5 mmHg in the NOVOS Core group
- Placebo: −1.5 mmHg
- Between-group difference: −6.1 mmHg (95% CI −10.9 to −4.9)
- The results exhibited statistical significance (p = 0.014)
Importantly, this population was not hypertensive. Baseline systolic blood pressure averaged in the mid-120s mmHg, already within the normal range.
How does NOVOS Core compare with other systolic blood pressure interventions?
In large meta-analyses of normotensive or generally healthy adults:
- Nitric oxide–focused supplements (beetroot/nitrate interventions) typically show reductions of ~2–4.4 mmHg.
- Aerobic exercise training often produces ~4 mmHg reductions.
- HIIT programs show similar modest reductions (3.9 mmHg) depending on baseline fitness.
- Resistance or isometric training generally produces ~2.9 mmHg reductions.
- Popular longevity ingredients such as quercetin, omega-3 fatty acids, and curcumin most often show average reductions in the ~2–2.6 mmHg range in non-hypertensive populations.
- Even well-structured dietary patterns such as the Mediterranean diet or DASH-style interventions frequently demonstrate modest reductions in normotensive adults (~1–3.9 mmHg).
Against that backdrop, NOVOS Core’s ~6 mmHg placebo-adjusted difference in generally healthy adults over six months is notable. Importantly, participants began the study within the normal blood pressure range, and systolic values remained within the normal range at follow-up. The observed change reflects support for healthy blood pressure already within the normal range, without inducing hypotension.
It places the magnitude of observed support for healthy systolic blood pressure already within the normal range at the upper end, and in most cases above what is typically reported for supplements or isolated lifestyle interventions studied in similar populations.
This does not imply treatment of hypertension or reduction in cardiovascular events. This trial was not designed or powered to measure clinical outcomes. However, from a physiological standpoint, the degree of blood pressure support observed here in a normotensive population is substantial relative to most non-pharmacologic interventions evaluated in comparable groups.

Simultaneous Multi-Endpoint Effects
One of the most significant aspects of this trial is not any single number, but that FMD, PWV, and SBP all moved favorably in the same randomized trial, without lipid modification.
Most nutritional studies:
- Move one endpoint modestly
- Or move lipids without affecting vascular function
- Or show acute but not sustained changes
Here, endothelial function improved, arterial flexibility increased, systolic pressure fell, and estimated SCORE2 risk shifted downward, all in parallel. That pattern is uncommon in supplement research.
Why Might This Be Happening?
We cannot claim mechanism from this trial alone. However, it’s worth emphasizing that NOVOS Core was not designed as a “blood pressure supplement” or “nitric oxide booster.”
It was designed around a hypothesis we filed a patent on in 2020: That simultaneously addressing multiple hallmarks of aging may influence downstream physiological systems more meaningfully than targeting a single pathway.
The NOVOS Core formulation was uniquely designed around the 12 biological hallmarks of aging and incorporates ingredients studied in peer-reviewed research for their roles in pathways linked to those hallmarks. These include redox balance, mitochondrial function, inflammatory signaling, and cellular stress response regulation –– interconnected biological processes that influence vascular aging and were relevant to the functional outcomes measured in this trial.
Importantly, the cardiovascular system was selected as the first human test case not because NOVOS was built for the heart alone, but because vascular aging is one of the earliest and most measurable manifestations of systemic aging biology. Our underlying hypothesis is broader: that targeting multiple hallmarks simultaneously may influence whole-body aging processes, with vascular function representing the first organ-level system evaluated in humans.
The manuscript discusses this multi-target rationale in detail. The vascular results are consistent with (though do not prove) the idea that addressing upstream biological aging mechanisms may manifest in organ-level functional improvements, as we’ve seen in this study.
Converging Lines of Evidence
This vascular trial doesn’t stand alone. Other data points in our broader research program include:
1. DunedinPACE Epigenetic Observational Investigation
In a 6-month observational cohort (n=12; 11 completers) taking NOVOS Core and NOVOS Boost (250 mg of nicotinamide mononucleotide), participants underwent epigenetic testing using the DunedinPACE algorithm before and after daily supplementation.
At follow-up:
- 73% of participants (8 of 11) demonstrated reductions in biological Pace of Aging beyond the test’s margin of error.
- The average reduction in Pace of Aging was −6.1%.
- The largest individual reduction was −14%.
- No participant showed an increase in Pace of Aging.
- The within-group change reached statistical significance (p = 0.001).

This investigation was exploratory, not placebo-controlled, and testing was conducted through a third-party laboratory. Epigenetic clocks are investigational tools and do not diagnose disease or predict lifespan. Larger, randomized controlled studies are necessary to confirm these findings.
Importantly, the direction of change in biological Pace of Aging was consistent with the functional vascular improvements observed in the NOVOS Core STAMINA randomized clinical trial. While the epigenetic findings cannot establish causality or clinical outcomes, the convergence between molecular aging metrics and organ-level physiological function provides a coherent signal across independent biological domains.
Taken together, these results suggest that the multi-pathway formulation strategy, designed around the 12 hallmarks of aging, may influence both upstream aging biology (epigenetics) and downstream functional physiology. Further controlled investigation is warranted.
2. Mouse Lifespan Study
In a naturally aged, diet-induced obese mouse model, continuous administration of the NOVOS Core formulation was associated with a statistically significant improvement in survival relative to controls. Median lifespan increased in the NOVOS Core group by more than 18%. The authors characterized this as a “rescue” from premature mortality under metabolic stress.
Mechanistically, the investigators hypothesized that the observed survival benefit may relate in part to senostatic effects (modulation of cellular senescence–associated pathways and inflammatory signaling) rather than direct senolytic activity. Supporting in vitro data demonstrated reductions in markers associated with genomic instability and cellular stress responses, consistent with engagement of aging-relevant biological processes.

NUT = NOVOS Core, SEN = R&D Rx senolytic combination (positive control), C = Control
“The study demonstrates that this multi-ingredient formulation exhibits synergistic effects comparable to pharmacological interventions, effectively compressing morbidity and maintaining cognitive function in late life [in mice].”
– Dr. Satomi Miwa, Translational Biology of Ageing, Newcastle University
While animal lifespan findings do not establish human longevity outcomes, the STAMINA randomized controlled trial demonstrates beneficial effects on validated markers of vascular aging in humans. Longer-term studies would be required to determine whether these physiological changes translate into clinical outcomes.
The study is currently under peer review, and the preprint is available here.
3. In Vitro Genomic Stability Research
Using advanced 3D human skin models, the NOVOS Core formulation demonstrated in a peer-reviewed study:
“A significant protective effect against genomic instability… a marked reduction in DNA damage markers and robust antioxidant activity, validating the formula’s potential to shield cells from a fundamental aging processes (DNA damage accumulation) and a process that is most probably implicated in healthspan (excessive oxidative stress).”
-Professor Antonello Lorenzini, Department of Biomedical and Neuromotor Sciences, University of Bologna
DNA damage accumulation and oxidative stress are established hallmarks of aging biology. The study administered chemotherapeutics to the human skin model and measured DNA strand breaks.
Cell models do not equate to clinical outcomes, but they help map out mechanisms.
4. Other
We have also conducted a small preliminary dermatological pilot (n=4) in which participants taking NOVOS Core for six months demonstrated objective improvements in skin firmness measured using a scientific indentometer, with improvements ranging from 12% to 40% (average ~22%). While exploratory and limited in size, these findings offer early clues that the formulation’s effects may extend beyond vascular physiology into other aging-relevant systems. Larger controlled dermatological studies are planned.
This is in addition to other in vitro studies finding favorable effects from the NOVOS Core formulation on aging-related mechanisms that include cellular senescence, cellular inflammation, and oxytosis/ferroptosis.
What This Does And Does Not Mean
This trial demonstrates that daily use of the NOVOS Core nutraceutical formulation was associated with:
- Improved endothelial function,
- Increased arterial flexibility,
- Lower systolic blood pressure, and
- A favorable directional shift in modeled 10-year cardiovascular risk estimates based on established population risk equations,
in generally healthy adults over six months.

Disclaimer: These findings do not demonstrate reduction in cardiovascular events, extension of human lifespan, or the treatment, prevention, or cure of any disease. Long-term, controlled outcome studies would be required to evaluate those endpoints.
A Shift in How Longevity Products Are Studied
Most consumer longevity products are supported primarily by ingredient-level citations. Among those that conduct original research, relatively few include animal lifespan studies (a foundational benchmark in biogerontology), and many human investigations rely on subjective surveys or molecular biomarkers rather than validated measures of organ-level physiological function.
University-conducted, randomized, placebo-controlled human trials measuring validated functional endpoints remain extremely uncommon in the consumer longevity space.
From aging biology and mechanistic research, to aged animal models, to randomized human evaluation of vascular function, the NOVOS program has now been studied end-to-end. That accomplishment matters as much as the results themselves.
“NOVOS take(s) a disciplined, biology-first approach in a space that too often prioritizes hype over evidence. What stood out to us was the seriousness of the scientific framework––mechanistic grounding, preclinical data, and an emerging human evidence base––combined with a willingness to engage in rigorous evaluation rather than shortcuts.”
– Dr. Doina Kulick, Lead Physician, Longevity Medicine at Mayo Clinic Arizona
If longevity science is to mature as a field, it must hold itself to higher evidentiary standards, measuring real physiology, in real humans, over meaningful durations. We believe this study represents a step in that direction.
Further investigation with larger trials, longer follow-up, and additional clinical endpoints will be essential. As NOVOS grows, we intend to continue expanding our research efforts.
For the first time, we can say that a multi-hallmark longevity formulation – NOVOS Core – was associated with significant, simultaneous improvements across three validated markers of vascular aging in a randomized controlled human trial, achieving some of the strongest results across each of these markers ever reported in a clinical study involving a generally healthy population.
That is a meaningful milestone, not just for NOVOS, but for the broader evolution of longevity science.
Table of Flow Mediated Dilation Comparable Studies
| Intervention | Sustained FMD improvement (%) | Population Studied | Study Type | NOVOS Improvement Advantage | Full Key Reference | Fasted? |
| NOVOS Core | 2.9 | Healthy adults ≥40 years | Randomized, double-blind, placebo-controlled trial | Reference | Yes | |
| Blood orange juice (dietary polyphenols) | 2.39 | Healthy adults with overweight and obesity | Randomized, controlled, single-blind, crossover trial | 1.21x | https://pubmed.ncbi.nlm.nih.gov/32510144/ | Yes |
| Tea (green/black tea) | 2.3 | Healthy adults | Meta-analysis of controlled human intervention studies | 1.26x | https://pmc.ncbi.nlm.nih.gov/articles/PMC3048861/ | Yes |
| Dietary Polyphenols | 2.22 | Mixed/at-risk (cardiometabolic risks) | Meta-analysis of RCTs | 1.31x | https://pubmed.ncbi.nlm.nih.gov/36796437/ | |
| Resistance training | 2.11 | Adults/older adults healthy | Meta-analysis of RCTs | 1.37x | https://pubmed.ncbi.nlm.nih.gov/34399984/ | NR |
| Blueberries | 2.01 | Healthy adults (subgroup; pooled RCTs) | Systematic review + meta-analysis of RCTs | 1.44x | https://pubmed.ncbi.nlm.nih.gov/38887319/ | |
| CoQ10 | 1.69 | Healthy subjects with mild-to-moderate dyslipidemia | Randomized, double-blind, placebo-controlled trial | 1.72x | https://pubmed.ncbi.nlm.nih.gov/32326664/ | Yes |
| Curcumin supplementation | 1.64 | Mixed/at-risk (cardiometabolic risks) | Meta-analysis of RCTs | 1.77x | https://pubmed.ncbi.nlm.nih.gov/39265778/ | |
| Coenzyme Q10 | 1.63 | Mixed/at-risk (cardiometabolic risks) | Meta-analysis of RCTs | 1.78x | https://pubmed.ncbi.nlm.nih.gov/38630421/ | |
| Nitric oxide booster | 1.6 | Healthy adults | Systematic review + meta-analysis of RCTs | 1.81x | https://pubmed.ncbi.nlm.nih.gov/40679494/ | NR |
| Folic acid | 1.51 | Healthy adults (no-CVD) | Systematic review + meta-analysis of RCTs | 1.92x | https://pubmed.ncbi.nlm.nih.gov/36829207/ | NR |
| Resveratrol | 1.43 | Adults (mixed populations across RCTs; not healthy-only) | Systematic review + meta-analysis of RCTs | 2.03x | https://pubmed.ncbi.nlm.nih.gov/35833325/ | |
| Resveratrol | 1.38 | Obese but otherwise healthy adults | Randomized, double-blind, placebo-controlled trial | 2.1x | https://pubmed.ncbi.nlm.nih.gov/23743811/ | Yes |
| Mediterranean diet | 1.3 | Middle-aged and older adults | Systematic review and meta-analysis | 2.23x | https://pubmed.ncbi.nlm.nih.gov/28424187/ | Yes |
| Aerobic exercise | 1.2 | Healthy adults ( normotensive ) | Meta-analysis of RCTs | 2.42x | https://pubmed.ncbi.nlm.nih.gov/40197331/ | NR |
| Cocoa | 1.2 | Healthy, middle-aged adults (35–60 years) | Randomized, double-blind, placebo-controlled trial | 2.42x | https://pmc.ncbi.nlm.nih.gov/articles/PMC4594054/ | Yes |
| Flavonoids | 1.16 | Adults (mixed populations across RCTs; not healthy-only) | Meta-analysis of RCTs | 2.5x | https://pubmed.ncbi.nlm.nih.gov/22301923/ | NR |
| Severe weight loss | 1.11 | Overweight/obese adults | Meta-analysis | 2.61x | https://pubmed.ncbi.nlm.nih.gov/25568949/ | Yes |
| Walnuts | 1.04 | Adults across mixed cardiometabolic profiles | Systematic review and meta-analysis of randomized controlled trials | 2.79x | https://pubmed.ncbi.nlm.nih.gov/40972869/ | NR |
| Flavan-3-ols | 1 | Not-elevated BP (<120/<70) (healthy-like) | Meta-analysis of RCTs | 2.9x | https://pubmed.ncbi.nlm.nih.gov/40126033/ | NR |
| Omega-3 | 0.95 | Without CHD, but with CHD risk factors | Systematic review + meta-analysis of RCTs | 3.05x | https://pubmed.ncbi.nlm.nih.gov/37552456/ | Yes |
Table of Pulse Wave Velocity Comparable Studies
| Intervention | PWV Improvement (m/s) | Study Duration | Population Studied | Comparator | NOVOS Improvement Advantage | Study Reference | Fasted? |
| NOVOS Core | 1.18 | 6 months | Healthy adults ≥40 years | Placebo | Reference | Yes | |
| DASH dietary pattern | 1.07 | 2 weeks | Overweight/obese unmedicated stage 1 hypertensive adults | Control diet | 1.1x | https://pmc.ncbi.nlm.nih.gov/articles/PMC3306995/ | Yes |
| Magnesium | 1 | 24 weeks | Overweight/slightly obese adults (45–70y), mostly healthy | Placebo | 1.18x | https://pubmed.ncbi.nlm.nih.gov/27053384/ | Yes |
| Omega-3 | 0.93 | 12 weeks | Healthy older adults | Pre–post | 1.27x | https://pubmed.ncbi.nlm.nih.gov/26109192/ | Yes |
| Severe weight loss | 0.8 | 3–12 months | Overweight/obese adults | Mixed (mostly pre–post; some controls) | 1.48x | https://pubmed.ncbi.nlm.nih.gov/25414255/ | NR |
| HIIT | 0.62 | 8–24 weeks | Adults with CVD risk factors/ at high risk for CVD | Control | 1.9x | https://pubmed.ncbi.nlm.nih.gov/38694567/ | NR |
| Nitric oxide booster (beetroot / nitrate) | 0.59 | 4 weeks | Hypertensive adults (18–85y) | Placebo | 2x | https://pubmed.ncbi.nlm.nih.gov/25421976/ | Yes |
| Aerobic exercise | 0.58 | 11 weeks (range 4–52) | Healthy Adults | Control | 2.03x | https://pubmed.ncbi.nlm.nih.gov/38101857/ | NR |
| Aerobic exercise (MICT) | 0.41 | ≥4 weeks (median 12; range 4–52) | Healthy Adults | Usual care | 2.88x | https://pubmed.ncbi.nlm.nih.gov/25333969/ | NR |
| Cocoa flavanols | 0.4 | 4 weeks | Healthy middle-aged (men and women (35–60y)) | Placebo | 2.95x | https://pubmed.ncbi.nlm.nih.gov/26348767/ | Yes |
| Vitamin K2 | 0.34 | 1 year | Healthy subjects 40–70y | Placebo | 3.47x | https://pmc.ncbi.nlm.nih.gov/articles/PMC11901762/ | NR |
| Mediterranean diet | 0 | 1 year | Healthy older adults, 65–79 years | Habitual diet | No improvement | https://pubmed.ncbi.nlm.nih.gov/30636547/ | NR |
Table of Systolic Blood Pressure Comparable Studies
| Intervention | SBP reduction used for comparison (mmHg) | Reported SBP range (mmHg) | Population | NOVOS Improvement Advantage | Full study reference | Fasted? |
| NOVOS Core | -6.1 | −10.9 to −4.9 | Healthy adults ≥40 years | Reference | Heiss C. , et al. A randomized, double-blind, placebo-controlled trial of a multi-ingredient nutritional intervention on vascular function and blood pressure in healthy adults aged 40+. Manuscript under peer review, University of Surrey, 2024. | Yes |
| Soy nuts | −5.0 | SBP <120 mmHg | Normotensive subgroup | 1.22x | https://pubmed.ncbi.nlm.nih.gov/17533209/ | Yes |
| Cocoa | -4.4 | -0.9 to −7.9 | Normotensive subgroup | 1.39x | https://pubmed.ncbi.nlm.nih.gov/19910929/ | Yes |
| Nitric oxide booster (beetroot / nitrate) | -4.4 | -2.8 to -5.9 | Adults, majority healthy participants | 1.39x | https://pubmed.ncbi.nlm.nih.gov/23596162/ | NR |
| Flavonoids | -4.14 | -5.95 to -2.32 | Adults (mixed populations across RCTs; not healthy-only) | 1.47x | https://pubmed.ncbi.nlm.nih.gov/22301923/ | NR |
| Aerobic exercise | −4.04 | −5.32 to −2.75 | Normotensive subgroup | 1.51x | https://pubmed.ncbi.nlm.nih.gov/11926784/ | NR |
| HIIT | −3.92 | −6.1 to −1.7 | Normal blood pressure subgroup (healthy-like) | 1.56x | https://pubmed.ncbi.nlm.nih.gov/37491419/ | NR |
| DASH diet | -3.9 | -6.0 to -1.8 | Healthy adults subgroup | 1.56x | https://pubmed.ncbi.nlm.nih.gov/32330233/ | NR |
| Vitamin C | -3.11 | −4.52 to −1.71 | Non-hypertensive trials (subgroup) | 1.96x | https://pubmed.ncbi.nlm.nih.gov/22492364/ | NR |
| Resistance training | −2.86 | −4.6 to −1.1 | Normal blood pressure subgroup (healthy-like) | 2.13x | https://pubmed.ncbi.nlm.nih.gov/37491419/ | NR |
| Magnesium | −2.78 | −5.22 a −0.34 | General normotensive population | 2.19x | https://pubmed.ncbi.nlm.nih.gov/39519450/ | NR |
| Quercetin | −2.57 | −4.17 to −0.96 | Normotensive subgroup | 2.37x | https://pubmed.ncbi.nlm.nih.gov/35948195/ | NR |
| Dietary sodium reduction | −2.42 | −3.56 to −1.29 | Normotensive individuals (healthy-like subgroup) | 2.52x | https://pubmed.ncbi.nlm.nih.gov/23558162/ | NR |
| Severe weigth loss | -2.4 | Overweight nonhypertensive persons | 2.54x | https://pubmed.ncbi.nlm.nih.gov/1669517/ | NR | |
| Omega-3 | -2.38 | −3.62 a −1.13 | SBP <130 mmHg (healthy-like) | 2.56x | https://pubmed.ncbi.nlm.nih.gov/35647665/ | NR |
| Tea | -2.37 | -3.82 to -0.91 | Healthy adults subgroup | 2.57x | https://pubmed.ncbi.nlm.nih.gov/25137341/ | NR |
| Soy protein | -2.27 | −3.77 to −0.76 | Normotensive subgroup | 2.69x | https://pubmed.ncbi.nlm.nih.gov/21342608/ | NR |
| Endurance training | −2.1 | −3.3 to −0.83 | Prehypertensive adults | 2.9x | https://pubmed.ncbi.nlm.nih.gov/23525435/ | NR |
| Potassium | −2.1 | −3.81 a −0.38 | Normotensive/healthy-like | 2.9x | https://pubmed.ncbi.nlm.nih.gov/39519450/ | NR |
| Pistachios | −2.04 | −4.10 to 0.01 | Healthy adults (subgroup) | 2.99x | https://pubmed.ncbi.nlm.nih.gov/33198823/ | NR |
| Almonds | -1.76 | -2.36 to -1.17 | Healthy adults (subgroup) | 3.47x | https://pubmed.ncbi.nlm.nih.gov/32444059/ | NR |
| Curcumin | -1.67 | −2.30 to −1.04 | Healthy subjects (subgroup) | 3.65x | https://pubmed.ncbi.nlm.nih.gov/38220376/ | NR |
| Walnuts | −1.29 | −1.42 to −1.16 | Healthy subjects (subgroup) | 4.73x | https://pubmed.ncbi.nlm.nih.gov/32510725/ | NR |
| Mediterranean diet | -1.1 | −2.0 to −0.1 | Healthy older adults (>64y) | 5.55x | https://pubmed.ncbi.nlm.nih.gov/28424187/ | Yes |
| Flavan-3-ols | -0.5 | −6.6 to 5.5 | Not-elevated BP (<120/<70) (healthy-like) | 1,220x | https://pubmed.ncbi.nlm.nih.gov/40126033/ | NR |
| Coffee | 2.4 | 1.0 to 3.7 | Mostly normotensive participants | Worsening | https://pubmed.ncbi.nlm.nih.gov/10024321/ | NR |